Metals and Oxygen Reactions
Year 7 Chemistry Interactive Lesson
Welcome to the Metals and Oxygen Reactions Lesson!
In this interactive lesson, you will learn about how different metals react with oxygen.
You'll explore:
- What happens when metals combine with oxygen
- The products formed during these reactions
- Why some metals react more readily than others
- Real-world applications of metal-oxygen reactions
This lesson includes interactive activities, a quiz, and a fun game to help you understand the concepts.
Your progress will be automatically saved, so you can return to where you left off.
Click "Next" to start the lesson, or use the menu bar above to navigate to a specific task.
Key Concepts: Metals and Oxygen Reactions
Metal Reactivity
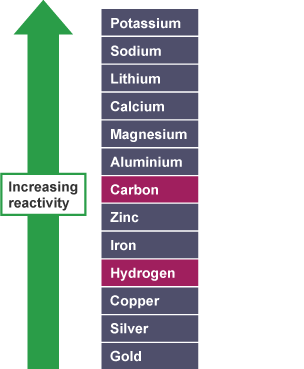
Different metals react with oxygen at different rates. Some metals, like potassium and sodium, are extremely reactive, while others like gold and platinum are very unreactive.
This difference in reactivity creates what we call the reactivity series - a way to rank metals from most to least reactive. Generally, the more reactive a metal is with oxygen, the more reactive it will be with other substances like water and acids.
Metal Oxides

When metals react with oxygen, they form metal oxides. The general equation is:
Metal + Oxygen → Metal Oxide
For example, when magnesium reacts with oxygen, it forms magnesium oxide:
Magnesium + Oxygen → Magnesium Oxide
Copper Patina: The Statue of Liberty

Did you know the Statue of Liberty was originally the color of a penny? It's made of copper, which naturally reacts with oxygen and carbon dioxide in moist air to form a greenish coating called patina.
This patina is a mixture of copper carbonate and copper hydroxide. It takes many years to develop fully, but once formed, it actually protects the copper underneath from further corrosion. This is why the Statue of Liberty has maintained its structure despite being exposed to the elements since 1886!
Aluminum's Protective Oxide Layer

Aluminum is actually a very reactive metal - more reactive than iron! However, aluminum doesn't rust like iron does. Why?
When aluminum reacts with oxygen, it forms a thin, transparent layer of aluminum oxide that tightly adheres to the surface. This oxide layer is protective - it prevents oxygen from reaching the aluminum underneath, stopping further oxidation.
This is why aluminum is widely used in applications like aircraft bodies, window frames, and food packaging, despite being highly reactive. It's an example of how understanding metal-oxygen reactions helps us choose the right materials for different applications.
Rusting: Iron Oxide Formation

When iron reacts with oxygen in the presence of water, it forms iron oxide, commonly known as rust. Unlike aluminum oxide, rust does not form a protective layer - it flakes off, exposing more iron to oxygen.
This is why iron objects need to be protected with paint, oil, or other coatings to prevent them from rusting. Without protection, an iron object left outside will eventually rust away completely!
Task 1: Match up!
Basic Knowledge
Match each term with the correct definition by clicking on a term and then on its matching definition.
Terms
Definitions
Task 2: Fill in the blanks
Basic Comprehension
Complete the paragraph below using words from the word bank:
When metals react with oxygen, they form metal . burns very in oxygen with a bright white flame. The of a metal tells us how easily it will react with oxygen. is an metal, which is why it stays shiny and doesn't tarnish. When iron reacts with oxygen and water, it becomes .
Task 3: Word Equations
Basic Application
Write word equations for these reactions of metals with oxygen. The first one is done for you.
1. Magnesium + Oxygen → Magnesium Oxide
2. Zinc + Oxygen →
3. Iron + Oxygen →
4. Copper + Oxygen →
5. Potassium + Oxygen →
Task 4: Ordering Reactivity
Medium Challenge
Using the information from your lesson, arrange these metals in order from MOST reactive with oxygen to LEAST reactive. Drag and drop the metals into the correct order.
Task 5: True or False?
Medium Challenge
Decide whether these statements are true or false. If false, rewrite the statement to make it correct.
1. All metals react with oxygen at the same rate.
2. Gold is unreactive and doesn't form an oxide layer.
3. Magnesium only reacts with oxygen when heated.
4. Metals that react vigorously with oxygen usually react vigorously with acids too.
5. Copper burns with a bright flame when heated in oxygen.
Task 6: Draw and Explain
Medium Application
Examine the stages of a magnesium ribbon burning in oxygen, then answer the questions below.
Stage 1: Before Burning
Magnesium appears as a shiny, silver-colored metal ribbon.
Stage 2: During Burning
Magnesium burns with an extremely bright white flame. The light is so intense it can damage eyes if viewed directly.
Stage 3: After Burning
The reaction produces white magnesium oxide powder. The reaction is: Magnesium + Oxygen → Magnesium Oxide
Describe what magnesium looks like before burning:
Describe what happens during the reaction:
Describe what the product (magnesium oxide) looks like:
Write the word equation for the reaction:
Task 7: Predict and Explain
Higher Challenge
For each scenario below, predict what will happen and explain your reasoning.
1. A gold ring is left outside for 10 years.
Prediction:
Explanation:
2. An iron nail is left in the garden for a month.
Prediction:
Explanation:
3. A copper roof is exposed to air for many years.
Prediction:
Explanation:
Task 8: Real-World Applications
Higher Challenge
Answer these questions about how we use our knowledge of metal reactions in everyday life.
1. Why are some bicycles made with aluminum frames rather than iron frames?
2. Why might gold be used for electrical connections in expensive audio equipment?
3. Why do we often paint or coat iron objects that will be used outdoors?
Task 9: Investigation Design
Advanced Application
Design a simple experiment to test which of three metals (iron, copper, and aluminum) is most reactive with oxygen.
What you would need:
The steps you would take:
What you would observe:
How you would record your results:
What safety precautions you would take:
Task 10: Creative Challenge
Advanced Application
Imagine you are designing a monument that needs to stand outside for 100 years without deteriorating.
1. Which metal(s) would you choose to use and why?
2. Which metal(s) would you avoid using and why?
3. What measures could you take to protect any less resistant metals that you might need to use?
Write a paragraph explaining your choices and reasoning:
Metals and Oxygen Quiz
Test your knowledge with this 10-question quiz about metals and oxygen reactions.
Question 1
What is formed when a metal reacts with oxygen?